Genomic DNA Extraction Kit (Whole Blood/Buffy coat)
No:DMGWB100 & 300
For research use only
Sample: up to 200 μl of whole blood, buffy coat, plasma, serum, body fluids or 5×106 lymphocytes
Yield : up to 50 μg
Introduction
This BioDiamond Genomic DNA Extraction Kit (Cultured Cell/Blood) was designed specifically for genomic DNA isolation from whole blood, frozen blood and buffy coat. Its unique buffer system ensures genomic DNA with high yield and good quality from samples while the spin column purifies and concentrates genomic DNA products previously isolated with the buffer system. The entire procedure can be completed in 1 hour without phenol/chloroform extraction needs. Purified genomic DNA is suitable for use in PCR or other enzymatic reactions.
Kit Contents
NOTE
★ Add ethanol (96–100%) to Buffer W2, shake before use (see bottle label for volume).
★ Add ddH2O to the proteinase K, vortex to dissolve and shake before use (see bottle label for volume).
★ Check Buffers before use for salt precipitation. Redissolve any precipitate by warming to 37°C.
★ Buffers W1 contain irritants. Wear gloves when handling these buffers.
Quality Control
In accordance with FairBiotech’s ISO-certified Total Quality Management System, the quality of the BioDiamond Genomic DNA Extraction Kit (Cultured cell/ Blood) tested on a lot-to-lot basis to ensure consistent product quality.
Additional requirements
*microcentrifuge tubes * absolute ethanol
*RNase A (10 mg/ml)
Protocol
Step 1 Sample Cells Harvesting
- Collect blood in EDTA-Na2 treated collection tubes (or other anticoagulant mixtures).
- Transfer up to 200 μl of blood or 200 μl of buffy coat to a sterile1.5 ml microcentrifuge tube.
- Add 20 µl of proteinase K and 200 µl of CK Buffer to the sample from Step 1 and mix by pulse-vortexing for 15 s.
- Incubate at 55ºC for 10 minutes. During incubation, invert the tube every 3 minutes.
Optional Step:
RNA Degradation (If RNA-free genomic DNA is required, perform this optional step.)
- Add 5 µl of RNase A (10 mg/ml) to the sample lysate and mix by vortex. Incubate at room temperature for 5 minutes.
- Add 350 µl of ethanol (96-100%) to the sample from Step 2 and mix by pulse-vortexing for 15 s.
- Place a CC Column in a 2 ml Collection Tube.
- Transfer the mixture completely to the CC Column.
- Centrifuge at 14,000 x g for 30 seconds.
- Discard the flow-through and place the CC Column back in the 2 ml Collection Tube.
- Add 400 µl of W1 Buffer into the CC Column. Centrifuge at 14,000 x g for 30 seconds.
- Discard the flow-through and place the CC Column back into the same Collection tube.
- Add 600 µl of W2 Buffer (Ethanol added) into the CC Column. Centrifuge at 14,000 x g for 30 seconds.
- Discard the flow-through and place the CC Column back into the same Collection tube.
- Centrifuge at 14,000 x g again for 2 minutes to remove residual W2 Buffer.
- Transfer the dried CC Column to a new 1.5 ml microcentrifuge tube.
- Add 50-200 µl of Pre-Heated EL Buffer or TE into the center of the column matrix.
- Let stand at 55ºC for 3 minutes.
- Centrifuge for 2 minutes at 14,000 x g to elute the purified DNA.
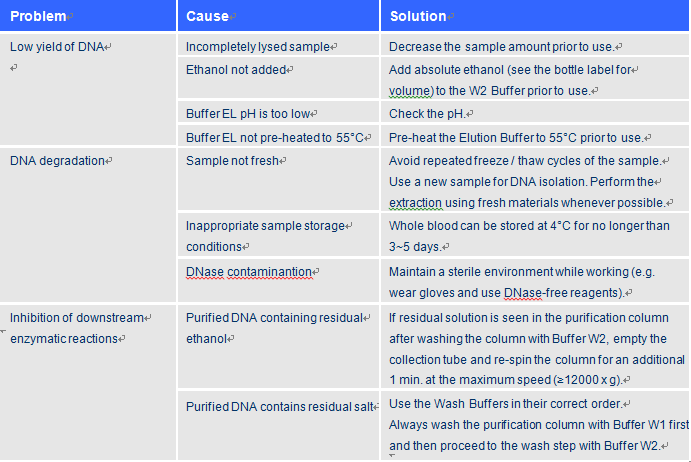
Troubleshooting
| dmgwb100___300.pdf | |
| File Size: | 247 kb |
| File Type: | |